Dimeric Collection
"Dimeric: Unveiling the Intricate World of Protein Interactions" Proteins are the building blocks of life
For sale as Licensed Images
Choose your image, Select your licence and Download the media
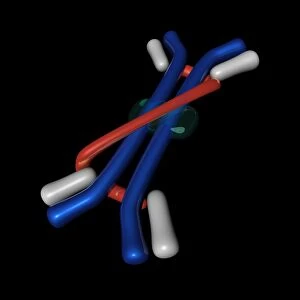
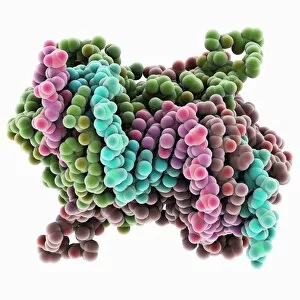
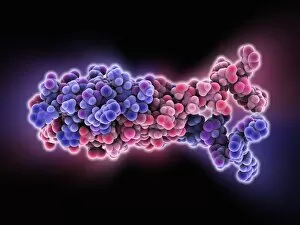
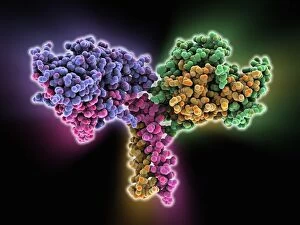
"Dimeric: Unveiling the Intricate World of Protein Interactions" Proteins are the building blocks of life, and their interactions play a crucial role in various biological processes. Among these interactions, dimerization stands out as a fascinating phenomenon that brings together two protein subunits to form a functional unit. In this captivating journey into the world of dimers, we explore some intriguing examples. Firstly, we encounter the Epstein-Barr virus protein bound to DNA C014/0875. This dimeric complex plays a pivotal role in viral replication and pathogenesis by manipulating host cell machinery. Moving on, our exploration takes us to Immunoglobulin A's molecular model—a remarkable example of dimerization in antibody structure. This process enhances its ability to neutralize pathogens and protect our immune system. Next up is the H-NS chromatin-structuring protein—an essential player in organizing bacterial DNA. Its dimeric form ensures proper gene regulation and genome stability. Continuing our expedition through diverse proteins, we stumble upon Newcastle disease virus protein C015/6912—a striking example of viral hijacking mechanisms involving dimer formation for efficient infection. The Kinesin motor protein dimers C015/5921 and C015/5920 captivate us with their role in intracellular transport along microtubules—essential for cellular functioning and division. Our journey then leads us to SMAD4 protein domains bound to DNA C015/6552 and C015/6551—key players in TGF-beta signaling pathway regulation. These dimers orchestrate intricate cellular responses critical for development and homeostasis. As we delve deeper into viral strategies, interferon antagonism by viral proteins C015/5421 and C015/5420 emerges as an intriguing mechanism employed by viruses to evade host immune defenses through disrupting antiviral responses mediated by interferons.